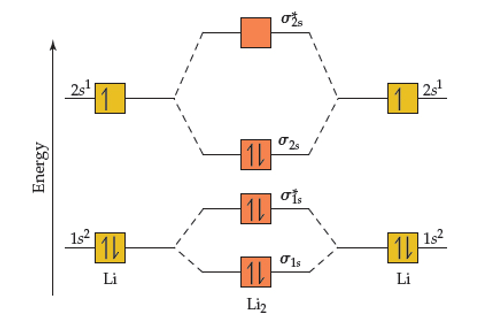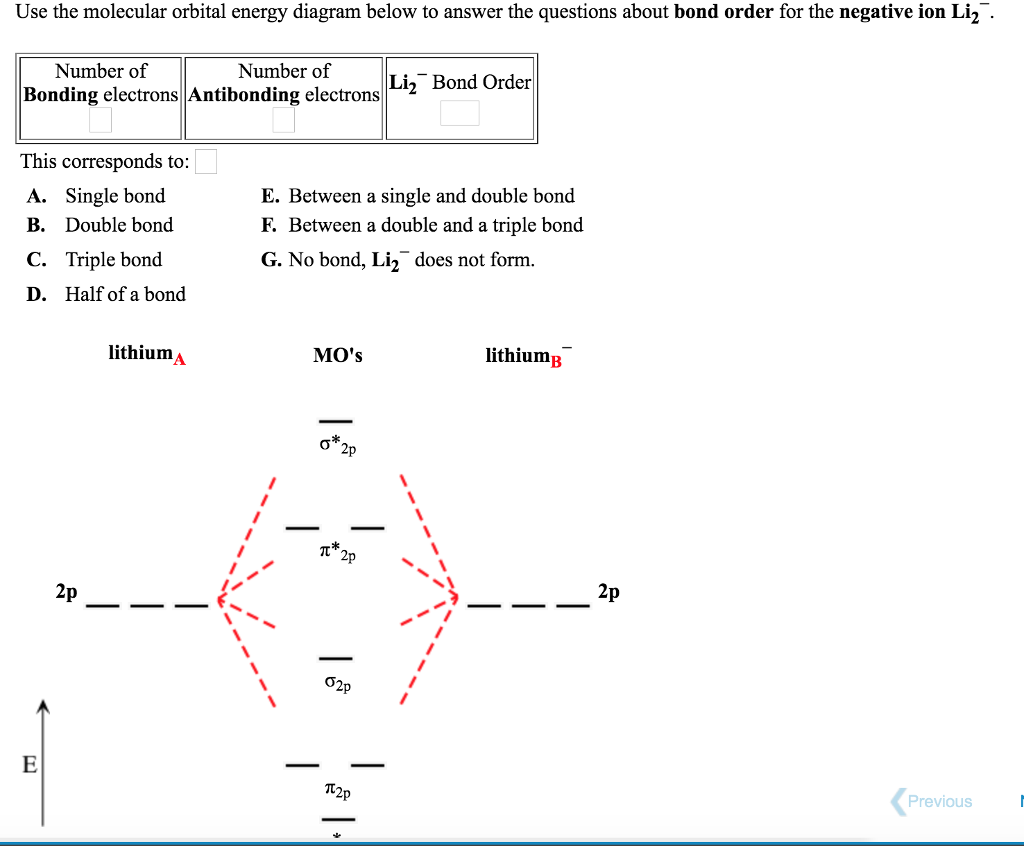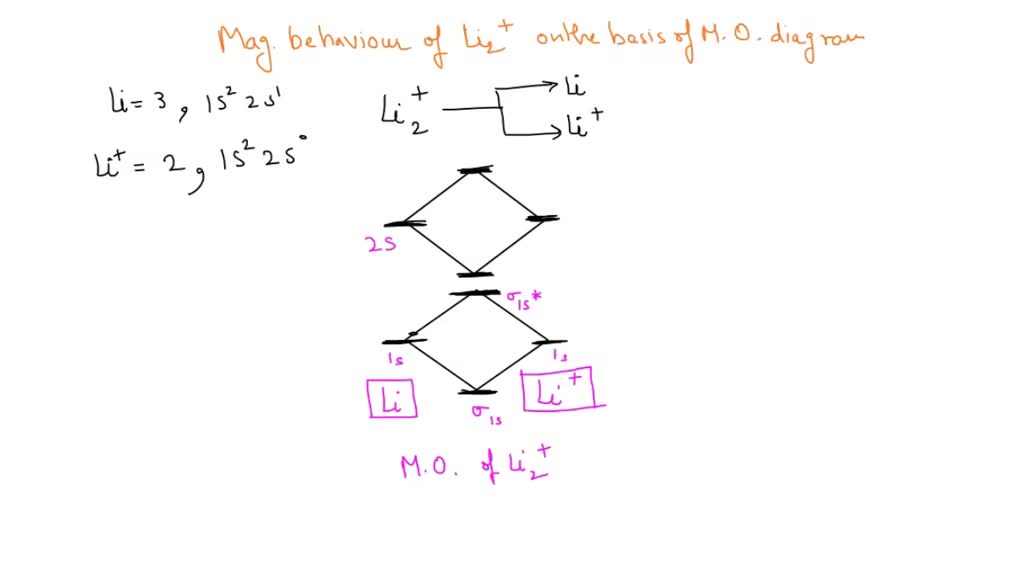
SOLVED: Based on the MO diagram of LI2+, predict if the compound is diamagnetic or paramagnetic. (The diagram for LI2 is similar to that of N2.)

Exploring response of Li2 molecule to external electric field: A DFT and SAC-CI study - ScienceDirect
According to molecular orbital theory, which of the following is true with respect to Li2+and Li2 ? 1)Both are unstable (2) Li2+ is unstable and Li2 is stable (3)Li2+ is stable and
Explain the formation of the following molecules on the basis of MOT. Also find the bond order. i. H2 ii. Li2 - Sarthaks eConnect | Largest Online Education Community