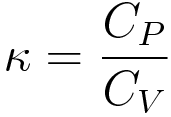OpenFOAM-9 multiphaseEulerFoam, wallBoiling, changing table Hs to accommodate new pressure : r/OpenFOAM

Are there any sort of incompatibilities between the Optimization Library of Modelica and the TIL commercial library? - Stack Overflow

Energy Conversion CHE 450/550. Ideal Gas Basics and Heat Capacities - I Ideal gas: – a theoretical gas composed of a set of non-interacting point particles. - ppt download

The equation is given below, CP=CV+TV(Beta2/K), links CP and CV with Beta and K. Use this equation to evaluate CP - CV for an ideal gas. CP=CV+T(Lambda P Lambda T)V(Lambda V Lambda

The Circle of Omicron Delta Kappa, Annual Report 2022, Vol. 101, No.1 by Associate Executive Director - Issuu